This EHR Module is 2014 compliant and has been certified* by an ONC-ACB in accordance with the applicable certification criteria adopted by the Secretary of the U.S. Department of Health and Human Services. This certification does not represent an endorsement by the U.S. Department of Health and Human Services or guarantee the receipt of incentive payments. DocuTrac, Inc. received ONC Modular EHR Ambulatory Certification on October 9, 2014 for QuicDoc Enterprise v8.1.0, Certification ID number 1314E01PO8QIEA1 and Drummond Group Certificate No. 09252014‐2912‐6.
This EHR Module is 2014 compliant and has been certified* by an ONC-ACB in accordance with the applicable certification criteria adopted by the Secretary of the U.S. Department of Health and Human Services. This certification does not represent an endorsement by the U.S. Department of Health and Human Services or guarantee the receipt of incentive payments. DocuTrac, Inc. received ONC Modular EHR Ambulatory Certification on October 9, 2014 for QuicDoc Enterprise v8.1.0, Certification ID number 1314E01PO8QIEA1 and Drummond Group Certificate No. 09252014‐2912‐6.
The purchase from DocuTrac, Inc. of QuicDoc Enterprise v8.1.0 will be delivered for a one-time per user fee and an annual support fee that will grant the user a perpetual license for v8.1.0 and encompass the base software. In addition, in order to attempt to meet meaningful use measures the end user of this certified product will be responsible for any cost incurred for the implementation and use of DrFirst Rcopia and MS HealthVault software, with the technical and contractual costs and limitations as described below.
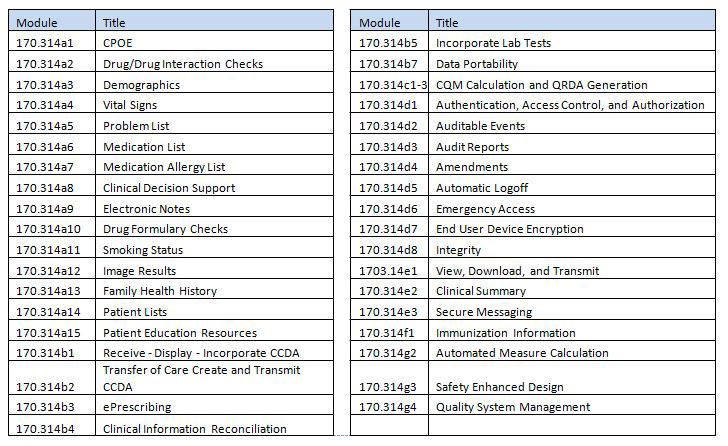
This certified version will require the end user to purchase the ePrescribing service (listed as additional software) with a one-time set up fee and annual fee per prescriber in order for the end user to become Meaningful Use compliant for CPOE, Drug/Drug Integration Checks, Drug Formulary Checks and Medication Allergy List. (170.314a1, 170.314a2, 170.314a10, 170.314a7)
This certified version may require ongoing monthly or annual fees to support an online patient service portal. Should the end user chose to use a patient portal other than HealthVault PHR, the end user must enter into a contract and pay the third party provider for the portal. DocuTrac, Inc. will set up the necessary data transfer mechanism to connect to that portal at no additional cost.(170.314e1)
To facilitate automatically integrated lab results this certified product will require an interface be established between this certified product and the identified lab. Support for the integration of these lab interfaces to facilitate the transfer of results may require additional one-time costs and annual support fees. (170.314b5)
When facilitating the exchange of information with Health Information Exchanges (HIE), Regional Health Information Organizations (RHIO), and any other data sharing entities this certified product may require the establishment of interfaces between the entities. Support for the integration of these interfaces to facilitate data transfer may require additional one-time costs and annual support fees. (170.314b7, 170.314e1)
To provide Secure Messaging from with-in this certified product there is a one-time set up fee and an annual support fee per messaging user. (170.314e3)